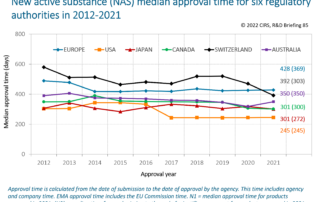
CIRS RD Briefing 94 – Value of Reference Agency Reports in Enabling Reliance
Access to information, including the assessment documents of reference national regulatory agencies (NRA), is a key enabler of regulatory risk-based decision making. It promotes an understanding of what was [...]