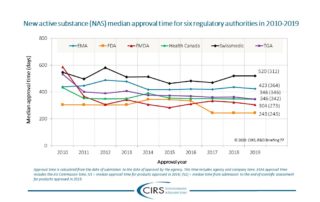
Bujar et al 2020 – Documentation of Quality Decision-Making Practices
Background: Pharmaceutical companies and regulatory agencies endeavor to relate their decision making with outcomes to improve future decision making and to ensure that gained knowledge is fed back into [...]