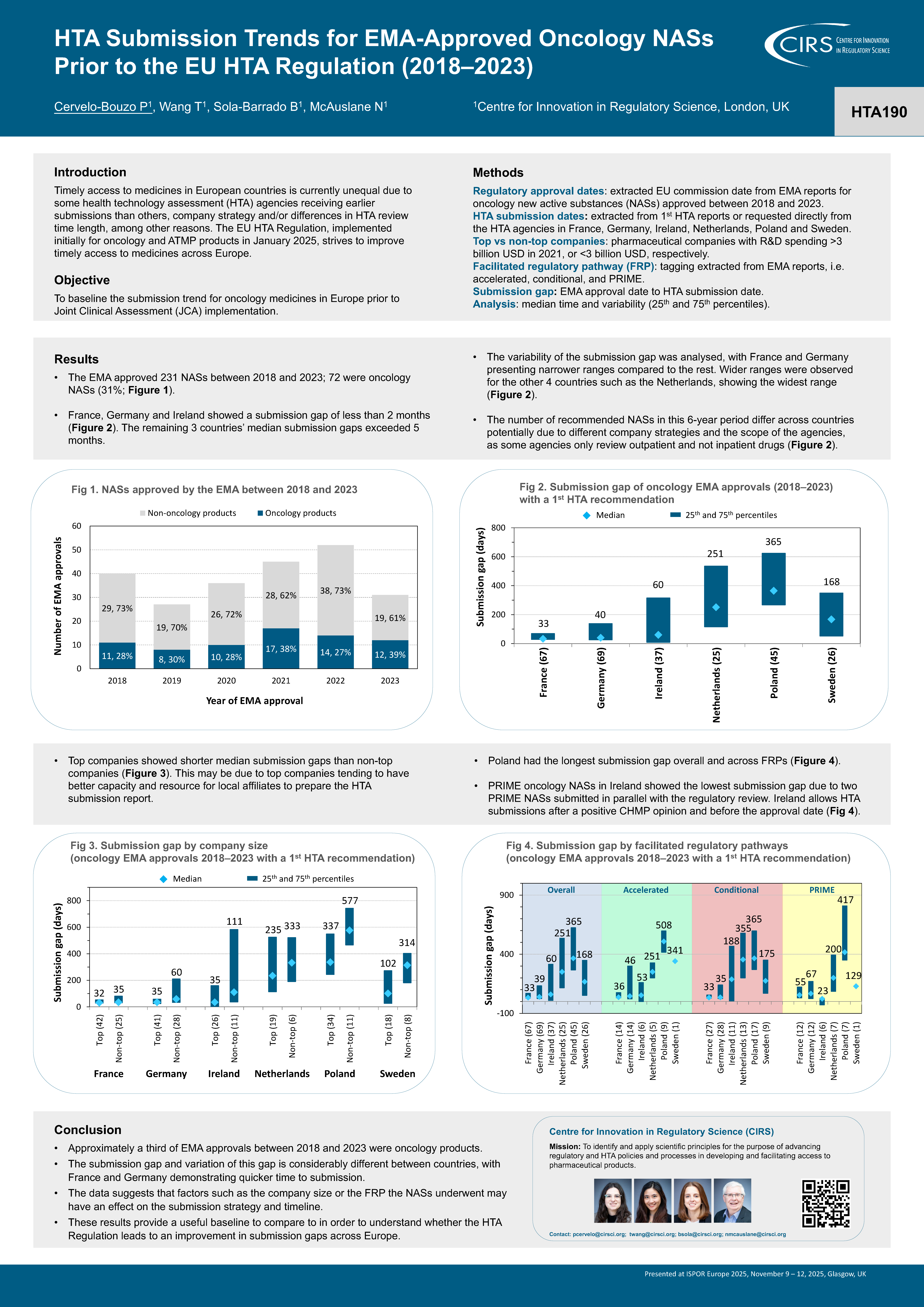
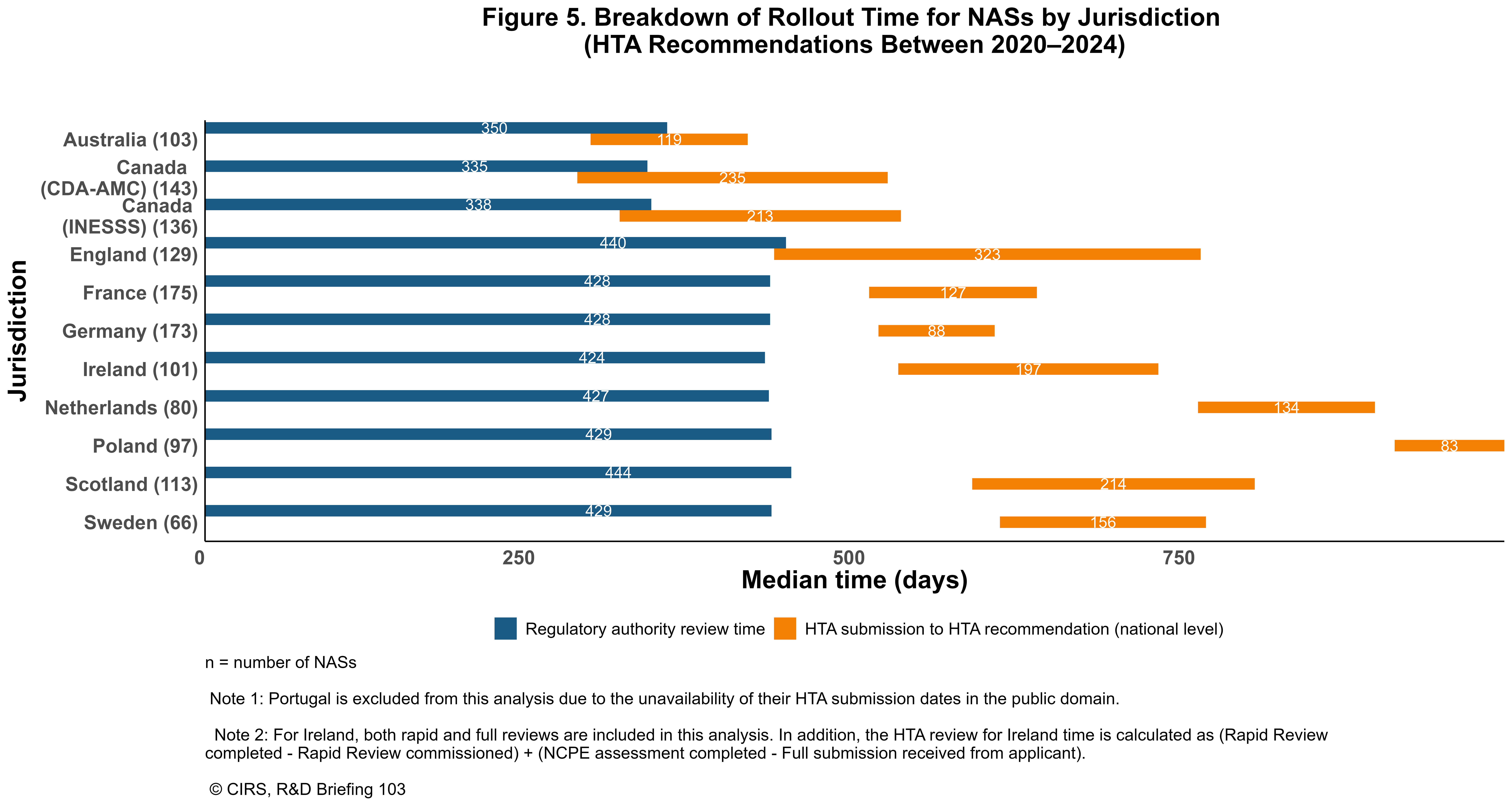
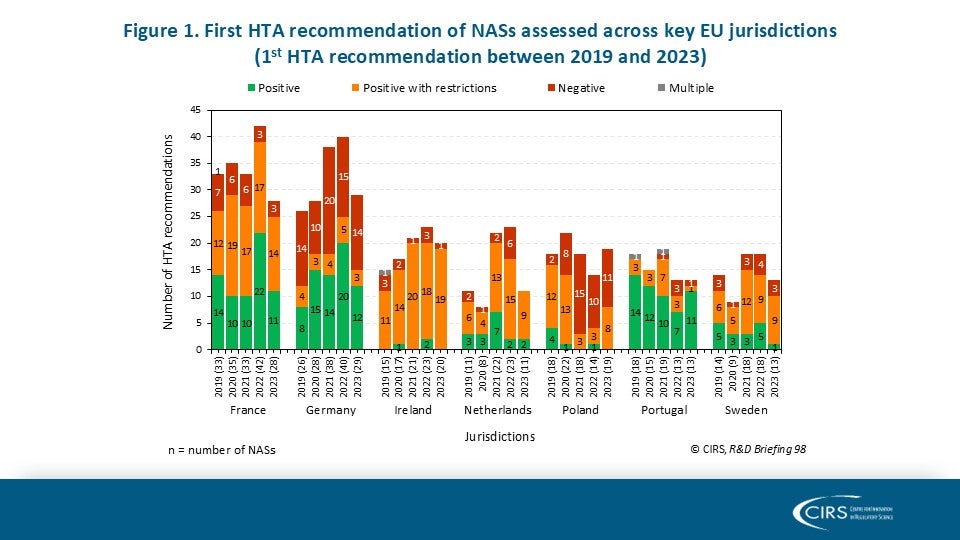
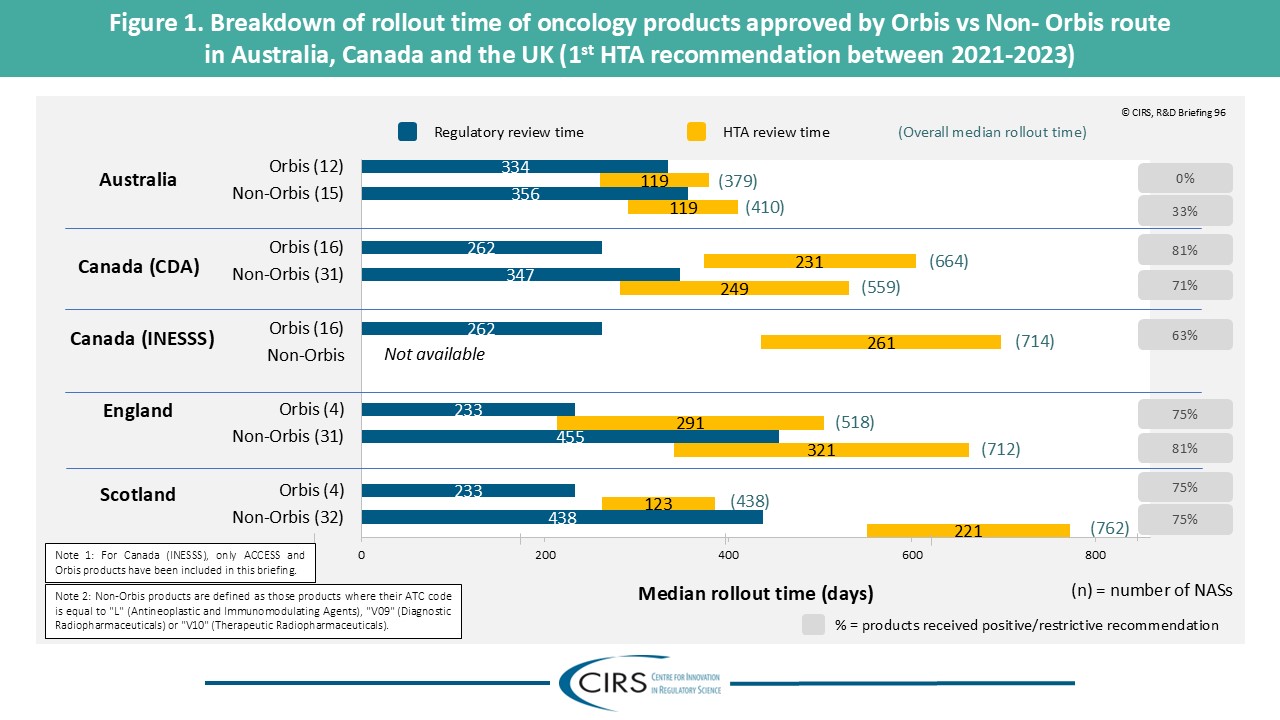
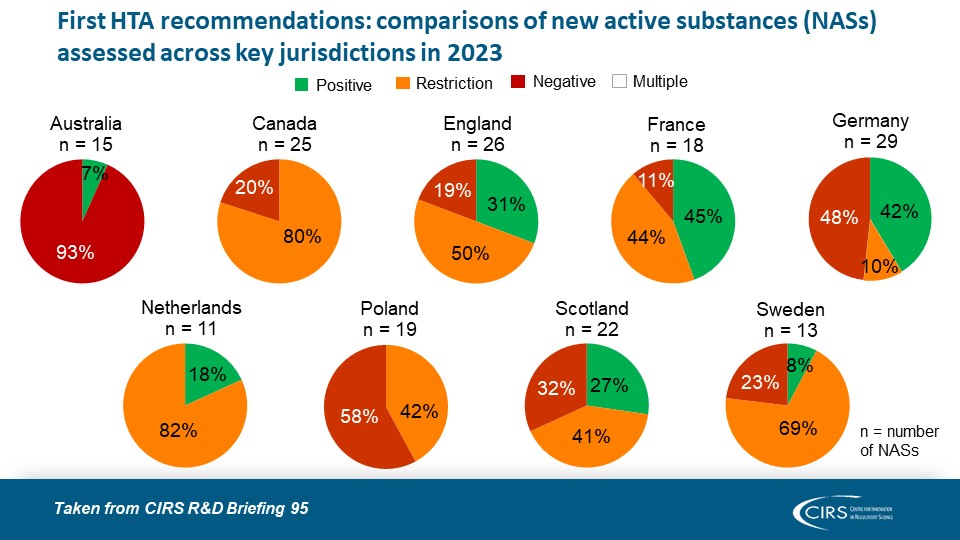
Parallel Submissions in Australia and Canada – Sola-Barrado 2026
As companies navigate diverse global HTA environments, new CIRS analysis provides timely evidence on the role of parallel regulatory/HTA submissions. Our latest publication in the International Journal of Technology [...]