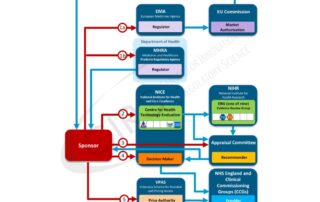
Liu et al 2013 – Characterising Good Review Practices across APEC agencies
As a first step in the implementation of the Asia-Pacific Economic Cooperation (APEC) Best Regulatory Practice Project, the Centre for Innovation in Regulatory Science conducted a gap analysis survey [...]