Publications
CIRS publishes insights from its research and meetings in several forms:
- R&D Briefings – research papers produced by the CIRS team e.g. annual regulatory and HTA benchmarking briefings
- Journal articles – peer reviewed academic research papers
- Reports – from CIRS workshops and externally commissioned research projects, as well as CIRS Annual Reports
- Books – research theses from CIRS-supported PhD students
- Posters – presented at external conferences
Keep up-to-date with CIRS publications and activities by signing up to our mailing list or following CIRS on LinkedIn.
2022 Workshop report – Building on regulatory and HTA agilities for high unmet need
In 2020, coronavirus disease (COVID-19) spread rapidly around the world and halted regular social, business, and research activities. The pandemic impeded normal functioning across several fields, and businesses and agencies …
2022 Workshop report – Collaborative models for regionalisation, work and information sharing: How do these fit into the regulatory toolkit?
This workshop looked at how maturing markets are building risk-based approaches into regulatory assessment, building on recent CIRS workshops in Singapore (2019) and South Africa (2018). The workshop also explored …
Regulatory, HTA and company interactions: the current landscape and future ecosystem for drug development, review and reimbursement
Background: Multi-stakeholder interactions have evolved at product and policy levels. There is a need to assess the current and future landscape of interactions between companies, and regulatory and HTA agencies to …
CIRS 2022 Annual Report
We’re delighted to present our latest Annual Report, which provides a summary of the projects, publications and Multi-stakeholder Workshops, Technical Fora, Industry Discussion Meetings, and Impact Case Studies from Regulatory …
2022 Workshop report – How has the pandemic accelerated the acceptance and utility of RWD/RWE in regulatory/HTA decision making?
This workshop builds on the outcomes of the CIRS Professor Breckenridge memorial workshop in December 2020, as well as the 2021 workshop on utilisation of digital technologies in clinical development. …
Evolution of HTA practice and approaches from the perspectives of HTA agencies and pharmaceutical industry
Health technology assessment (HTA) has emerged as an important tool to support healthcare decision-makers to make rational reimbursement decisions, with the ultimate purpose of promoting an efficient healthcare system. HTA …
2021 Workshop report – Regulatory and HTA landscape in Asia and Latin America
As regulatory systems mature with demand for new innovative treatments, jurisdictions are seeking to introduce more comprehensive healthcare systems; this is often accompanied by efforts to initiate health technology assessment …
Assessment of the effectiveness and efficiency of the West Africa medicines regulatory harmonization initiative by the member countries
Background: The West Africa Health Organization launched the West Africa Medicines Regulatory Harmonization Project (WA-MRH) in 2017 with the overarching objective to improve the availability of high-quality, safe and effective …
Evaluation of the Food and Drugs Authority, Ghana Regulatory Review Process: Challenges and Opportunities – Owusu‑Asante 2022
Purpose: This study aimed to assess the current regulatory review process of the food and drugs authority (FDA) Ghana by identifying key milestones, target timelines, good review practices and quality …
Evaluation of the East African Community joint assessment procedure by pharmaceutical companies
Background: A 2021 study to determine the viewpoints among the seven member countries regarding the effectiveness (i.e., achieving the intended outcomes) and efficiency (i.e., achieving the intended outcomes in timely …
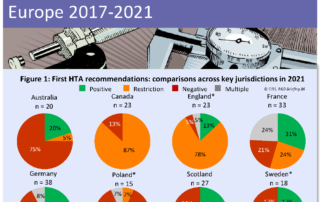
CIRS RD Briefing 86 – Review of HTA outcomes and timelines in Australia, Canada and Europe 2017-2021
The Briefing presents data from HTADock, an ongoing metrics study that collects data on new active substances (NASs) appraised by eight HTA agencies and analyses synchronisation between the regulatory decision …
Regulatory work-sharing initiative in Africa: ZaZiBoNa, past, present and future
PREFACE: The role of regulatory authorities in the health system is to ensure the quality, safety and efficacy of medical products. It is acknowledged that regulatory authorities are at times …
Evaluation of the East African Community joint assessment
Background: For almost a decade, the East African Community has implemented the Medicines Regulatory Harmonization (EAC-MRH) programme among its member states to harmonise technical requirements and standards for medical products …
Building HTA/Payer Perspectives Into Drug Development
Background: The target product profile (TPP) outlines the desired profile of a target product aimed at a particular disease and is used by companies to plan clinical development. Considering the increasing …
Evaluation of the performance of the Gulf centralised registration procedure
Background: The Gulf Centralised Committee for Drug Registration (GCC-DR), as part of the Gulf Health Council (GHC), enables the consolidated registration of pharmaceutical products throughout the member states of the …