This R&D Briefing presents the results from the Centre for Innovation in Regulatory Science (CIRS) annual analysis of new active substance (NAS) approvals by six major regulatory agencies: the European Medicines Agency (EMA), the US Food and Drug Administration (FDA), the Japan Pharmaceuticals and Medical Devices Agency (PMDA), Health Canada, Swissmedic and the Australian Therapeutic Goods Administration (TGA). The analysis focuses on 2023 as well as looking back at 2014-2023.
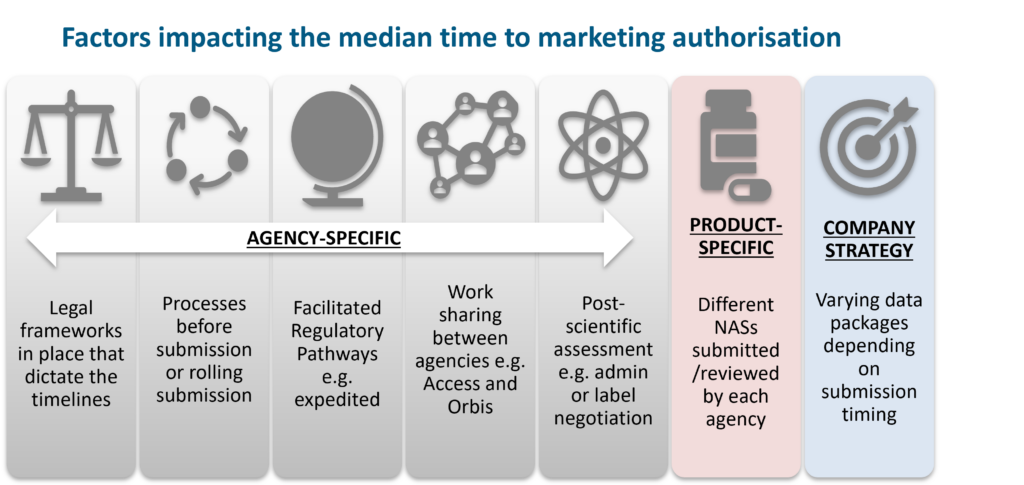
Although median approval times can be a marker of agency performance and the time it takes to make medicines available to patients, other factors must be considered, as illustrated below.
The NAS list associated with this R&D Briefing is available to download here.
Please cite this article as: Centre for Innovation in Regulatory Science (2024) R&D Briefing 93: New drug approvals in six major authorities 2014–2023: Changing regulatory landscape and facilitated regulatory pathways. Centre for Innovation in Regulatory Science (CIRS), London, UK.
Questions?
If you have any questions or comments on this study, please get in touch with Dr Magda Bujar: mbujar@cirsci.org

