As pharmaceutical innovation becomes increasingly global, companies are pursuing multi-jurisdictional approvals and timely patient access through evolving regulatory and reimbursement pathways. In China, regulatory reforms and expanded use of expedited pathways have contributed to more efficient drug approvals, alongside more dynamic approaches to reimbursement decision making.
This new CIRS briefing focuses on Class 1 innovative medicines approved in China between 2022–2024, defined as products not previously approved in China or overseas at the time of submission to the National Medical Products Administration (NMPA). These novel therapies provide an important indicator of innovation within China’s pharmaceutical ecosystem.
This study explored regulatory pathways, approval sequencing, and reimbursement outcomes, with comparisons to major global markets.
Key insights from the briefing:
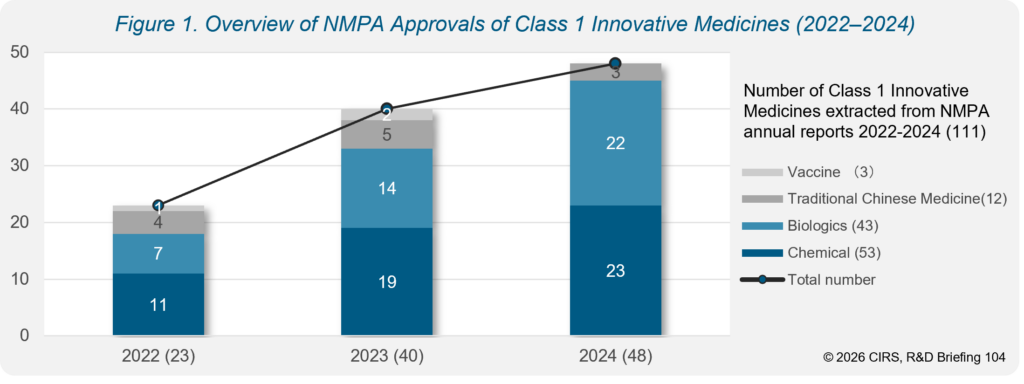
- Class 1 innovative medicine approvals more than doubled from 2022 to 2024 (from 23 to 48 approvals).
- Oncology was the leading therapeutic area, accounting for 46% of Class 1 approvals.
- Class 1 approvals were largely driven by domestic companies (79 vs 17 by multi-national companies).
- FDA was most often the first submission and approval jurisdiction for multi-national companies, with China typically included as an early, but not first, market (most NMPA submissions occurred within 31–180 days of the initial global filing).
- Use of expedited pathways increased over time, with priority review more common among multinational companies and conditional approval more frequently used by domestic companies.
- 66% of approved Class 1 medicines were included in China’s National Reimbursement Drug List (NRDL), with a median time of 360 days from regulatory approval to reimbursement.
This briefing highlights China’s evolving role in global pharmaceutical innovation, including the growing contribution of domestic innovation and the increasing importance of China in global development strategies.
Please cite this article as: Wang T, Cervelo P, Bujar M & McAuslane N (2025) R&D Briefing 102: Class 1 Innovative Medicines Approved in China (2022-2024): An International Comparison. Centre for Innovation in Regulatory Science (CIRS), London, UK.
Questions?
If you have any questions or comments on this study, please get in touch with Dr Tina Wang: twang@cirsci.org.

